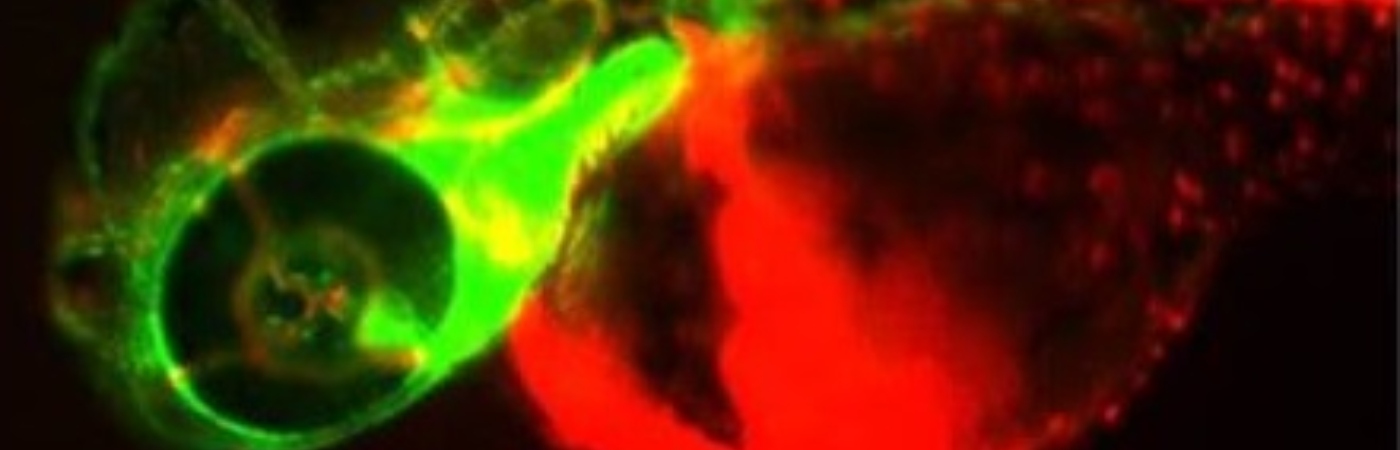
Dr Paul Kasher from The University of Manchester has developed a method of mimicking haemorrhagic strokes in zebrafish embryos, creating a more ethical model for laboratory research.
Haemorrhagic strokes account for 10 – 15% of strokes in the UK, and are responsible for approximately 6% of all global deaths. There are no specific drug treatments available for haemorrhagic stroke patients, a statistic which Dr Paul Kasher, a researcher specialising in stroke modelling, aims to improve through the use of zebrafish in research.
Currently, rodents – usually mice – are the most common animal species to be found in laboratories investigating strokes, and have been extremely useful for unravelling the biological mechanisms associated with bleeding in the brain. However, aside for the obvious ethical complications this throws up, Dr Kasher explains that the use of mice has not been particularly successful in terms of developing new drugs for haemorrhagic stroke patients.
“To create a haemorrhagic stroke in mice,” says Dr Kasher.“You have to surgically induce a bleed in the brain. This doesn’t necessarily mimic the spontaneous nature of blood vessel rupture that we observe in human haemorrhagic stroke.”
On the contrary, Dr Kasher’s zebrafish models, which he has developed with funding from NC3Rs, bleed in a manner which more closely mimics the spontaneity of the human condition. Through a combination of genetic editing and chemicals absorbed through the skin, researchers can induce a stroke in two-day old fish.
We hope a knock-on effect is to reduce the numbers of protected animals, such as mice, required for this type of research.
Dr Paul Kasher / Stroke Association HRH The Princess Margaret Lecturer
What’s more, zebrafish embryos are transparent, removing the need for invasive procedures to investigate the effects of the bleeding within the brain. This also allows researchers to watch the stroke in real time, shining a light on how the cells around the bleed respond.
“In making these models, we had two initial questions to answer,” says Dr Kasher. “The first was whether we could reduce the reliance on mammalian models, and the second was could we use fish for things we can’t do in rodents?
“For both points, the answer was yes.”
Real-time and at scale
As well as their transparency providing a real-time readout, the way the fish swam also helped to model the effects of a stroke – as an indicator of a physical disability caused by the stroke. Furthermore, the small size of the embryos, coupled with comparatively large numbers and short reproductive cycles, means fish models can be used to screen hundreds of drugs at a time.
Dr Kasher has recently received funding from the Stroke Association to utilise these advantages to identify drugs which might be capable of reducing brain injury post-stroke.
“At the moment, we are not focusing on trying to prevent the stroke,” he explains. “Instead, we’re assuming that the stroke will occur and we’re interested in how we can decrease the massive inflammatory response post-stroke, which is what exacerbates the injury.”
The best thing about this? It’s a happy ending for the fish, too. After suffering a stroke at two days (an age when zebrafish are not a protected species), the majority of the fish completely recover, and go on to produce the next generation of animals.
“The priority of this research is to contribute to our knowledge of haemorrhagic strokes,” Dr Kasher notes.
“But we hope a knock-on effect is to reduce the numbers of protected animals, such as mice, required for this type of research.”
